Any content you receive is for information purposes only. Always conduct your own research. *Sponsored
Krypton Street Just Added (Nasdaq: MBRX) to the Watchlist This Morning — Wednesday, May 13, 2026 Don’t Miss The Next Breakout—Get Real-Time Alerts Sent Directly To Your Phone. Up To 10X Faster Than Email.
(MBRX) Full Coverage Starts Now
May 13, 2026
First Look—Here's Why (Nasdaq: MBRX) is the Only Ticker We’re Pull Up Today
Dear Reader, Krypton Street is tracking a little-known Phase 3 biotech that could be approaching one of its most important clinical milestones yet. Preliminary blinded data from the first 30 subjects showed a 40% composite complete remission rate, approximately 67% above the historical response rate for standard cytarabine alone, while management has publicly stated interim Phase 3 data is expected later this quarter. That is part of the reason why Moleculin Biotech, Inc. (Nasdaq: MBRX) just hit our radar and is climbing toward the top of this morning’s watchlist—Wednesday, May 13, 2026. But keep in mind, (MBRX) has less than 6M shares listed as available to the public. When companies have small public floats like this, the potential exists for big moves if demand begins to shift. Recently, (MBRX) made an approximate 50% move in just over a month when it went from around $1.79 on March 9 to $2.72 on April 14, according to Barchart. That kind of setup is exactly what can put a little-known biotech under a much brighter spotlight in a very short window. And with a pivotal clinical update now expected before quarter-end, (MBRX) could start to draw attention from more than just our desk. Analyst Targets Point To 300%–800% Upside Potential…
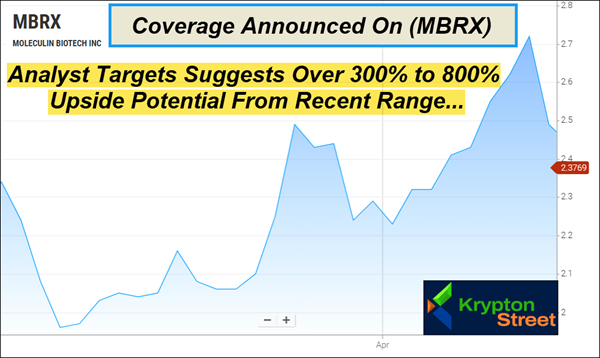
Sara Nik of H.C. Wainwright has a $22 target on (MBRX), which suggests over 800% upside potential from its recent $2.40 range. Jason McCarthy of Maxim Group reiterated a $10 target less than two weeks ago, which suggests over 300% upside potential from current levels. Two covering analysts, two significant targets, one imminent data readout. (MBRX) ended 2025 with $8.9M in cash and raised an additional $8.3M in gross proceeds through financing activities in Q1 2026. Management has confirmed it expects combined resources to fund planned operations into Q3 2026 — through and beyond the anticipated MIRACLE interim readout. But the analyst targets and near-term clinical timing are only part of the story. To understand why (MBRX) is drawing this level of attention, you have to look at the science behind the pipeline itself. A Pipeline Built for Cancers That Fight Back
Moleculin Biotech, Inc. (Nasdaq: MBRX) is a Houston-based, late-stage pharmaceutical company developing therapeutic candidates for hard-to-treat tumors and viruses. Its lead program, Annamycin (naxtarubicin), is a next-generation anthracycline engineered with two critical advantages over older agents like doxorubicin: it is designed to avoid multidrug resistance mechanisms, and it lacks the cardiotoxicity that has long constrained the use of conventional anthracyclines. Across more than 100 patients treated in Moleculin's clinical programs to date, no associated cardiotoxicity has been reported — a documented safety differentiator that could matter considerably in regulatory and commercial discussions. 
Annamycin is being evaluated in the MIRACLE trial — a pivotal, global Phase 2B/3 study combining Annamycin with cytarabine (AnnAraC) for adult R/R AML patients following induction therapy. The trial is enrolling across eight countries in the United States and Europe. The company has also completed a successful Phase 1B/2 study (MB-106) that, with FDA input, it believes has substantially de-risked the regulatory pathway toward a potential new drug approval. Beyond AML, the pipeline includes Annamycin for soft tissue sarcoma lung metastases; WP1066, an immune and transcription modulator targeting brain tumors and other cancers; and WP1122, an antimetabolite being explored for viral indications. (MBRX) is a multi-indication platform with AML leading the charge. The Market Moleculin Is Targeting
The global AML treatment market was valued at approximately $3.87B in 2025 and is projected to expand at a CAGR of roughly 10.6%, reaching an estimated $9.62B by 2034. 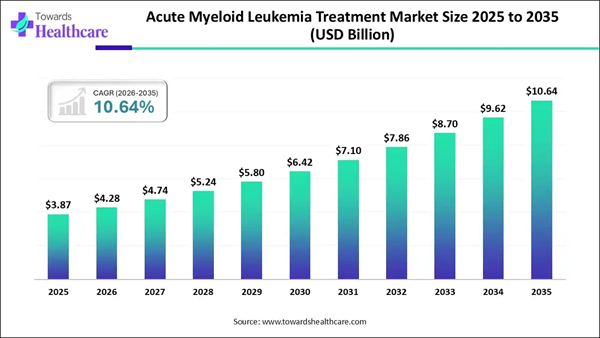
The American Cancer Society estimates 22,010 new AML diagnoses in the United States in 2025 alone, with approximately 11,090 expected deaths. For the relapsed or refractory subset — Moleculin's specific target population — the unmet need is especially severe: standard high-dose cytarabine (HiDAC) produces complete remission rates of only around 17-18%, leaving the vast majority of patients without durable responses. That is the gap AnnAraC is designed to fill. The preliminary blinded 40% composite complete remission rate observed across the first 30 MIRACLE subjects — of whom approximately 35% had already failed prior venetoclax-based therapy, among the most resistant patients — supports the view that AnnAraC may be performing meaningfully above the standard of care. Full unblinded confirmation is expected this quarter. A Cascade of Recent Potential Catalysts
On May 8, 2026, Moleculin announced a new Hong Kong patent (No. 40073244) covering its proprietary method of reconstituting liposomal Annamycin, extending IP protection through June 25, 2040. The timing is deliberate. CEO Walter Klemp stated directly: "This level of patent protection is made all the more important given the expected timing of our first Phase 3 clinical data release later this quarter." (MBRX)’s global IP now spans four continents. On April 23, new preclinical data presented at the AACR Annual Meeting 2026 showed Annamycin extending survival by more than 60% in metastatic pancreatic cancer preclinical models, with significantly greater tumor tissue accumulation than doxorubicin (p<0.0001), and evidence of CD8+ and CD4+ T cell infiltration — suggesting potential to activate immune responses in notoriously cold tumors. An abstract highlighting Annamycin data has also been accepted for poster presentation at the 2026 ASCO Annual Meeting, one of oncology's most influential annual gatherings, placing Annamycin in front of a global clinical audience at exactly the right moment. May 13, 2026: 7 Reasons Why (MBRX) Has Earned The Top Spot On Our Watchlist This Morning…
1. Recent Momentum: After moving from around $1.79 on March 9 to $2.72 on April 14, (MBRX) already showed an approximate 50% move in just over a month. 2. Small Float: With fewer than 6M shares listed as available to the public, (MBRX)’s small float could witness the potential for big moves if demand begins to shift. 3. Analyst Targets: With targets of $10 and $22, (MBRX) has analyst coverage which suggests over 300% to 800% upside potential from its recent $2.40 range. 4. Clinical Timing: With MIRACLE interim data expected later this quarter, (MBRX) has a near-term readout that could draw added attention. 5. Early Signal: Based on preliminary blinded results from the first 30 subjects, (MBRX) showed a 40% composite complete remission rate. 6. Patent Coverage: Following a new Hong Kong patent extending protection through June 25, 2040, (MBRX) now has global IP spanning four continents. 7. Capital Position: After ending 2025 with $8.9M in cash and raising another $8.3M in Q1 2026, (MBRX) says resources should fund planned operations into Q3 2026. Get (MBRX) On Your Radar While It’s Still Early…

Taken together, the setup surrounding (MBRX) is difficult to ignore right now. Between the small public float, the recent approximate 50% move in just over a month, analyst targets ranging from $10 to $22, and a pivotal MIRACLE interim data readout expected later this quarter, multiple factors are beginning to converge at the same time. Add in the preliminary 40% composite complete remission rate, capital resources projected into Q3 2026, and expanding patent protection across four continents, and it becomes clear why (MBRX) just hit our radar. (MBRX) has our full attention this morning. Take a look at (MBRX) before the opening bell. My next update could be on the way shortly, so keep watch. Sincerely, Alex Ramsay
Co-Founder / Managing Editor Krypton Street Newsletter
|





