Any content you receive is for information purposes only. Always conduct your own research. *Sponsored
GT Biopharma, Inc. (Nasdaq: GTBP) Headlines The Market Crux Watchlist Tomorrow Morning— Thursday, May 14, 2026
Don’t Miss The Next Breakout—Get Real-Time Alerts Sent Directly To Your Phone. Up To 10X Faster Than Email.
We’ll Be Opening (GTBP) Coverage Early
Don’t Leave (GTBP) Off Tomorrow’s Watchlist…
May 13, 2026
Tonight’s Preview | (Nasdaq: GTBP) Coverage is Dropping Early — Don't Sleep on This
Dear Reader, Market Crux has identified a clinical-stage biotech below $0.50 that recently made an approximate 59% move in under a week while still flying well under the radar. 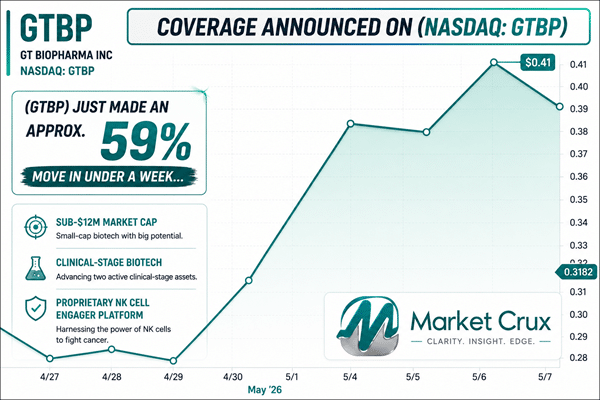
GT Biopharma, Inc. (Nasdaq: GTBP) is developing a proprietary NK cell engager platform designed to activate the body’s natural killer cells directly in vivo, with clinical programs already underway and additional studies approaching. Backed by two active clinical-stage assets, a growing pipeline, and several defined clinical timelines extending through 2026, (GTBP) quickly became one of the primary names on the Market Crux watchlist ahead of Thursday morning, May 14, 2026. What makes this setup stand out is the combination of near-term clinical timelines, recent momentum, and a platform aimed at several difficult-to-treat disease areas. For a company still flying this far under the radar, (GTBP) gives readers plenty to pay attention to as Thursday’s session approaches.
How GT Biopharma, Inc. (Nasdaq: GTBP) Targets
Cancer Differently
GT Biopharma, Inc. (Nasdaq: GTBP) is a San Francisco-based clinical-stage biopharmaceutical company developing immuno-oncology therapeutics based on its proprietary TriKE® (Tri-Specific NK Cell Engager) platform. Unlike CAR-T therapies — which require cells to be harvested and modified outside the body — (GTBP)’s approach is designed to harness and enhance the cancer-killing ability of a patient's own NK cells directly in vivo, without removing or modifying those cells prior to treatment. 
The company holds an exclusive worldwide license agreement with the University of Minnesota to develop and commercialize therapies using the TriKE® technology. The TriKE® platform is built around a tri-specific molecule with three functional domains: an anti-CD16 nanobody that binds and activates NK cells via antibody-dependent cellular cytotoxicity (ADCC), an IL-15 crosslinker that promotes NK cell expansion and persistence at the tumor site, and an anti-tumor associated antigen (TAA) binding domain that directs NK cells to specific cancer markers. This modular architecture allows (GTBP) to efficiently adapt the platform across different tumor targets — a key design advantage. (GTBP)’s active pipeline includes three named TriKE® candidates: GTB-3650, a second-generation camelid nanobody TriKE® currently in a Phase 1 dose escalation trial for relapsed or refractory (r/r) CD33-expressing hematologic malignancies including acute myeloid leukemia (AML) and high-risk myelodysplastic syndrome (MDS); GTB-5550, a B7-H3-targeted TriKE® that received FDA IND clearance in February 2026 with a Phase 1 basket trial planned for mid-2026; and GTB-7550, a CD19-targeted candidate in preclinical development for lupus and other autoimmune disorders. The $136B Immunotherapy Backdrop Behind (GTBP)
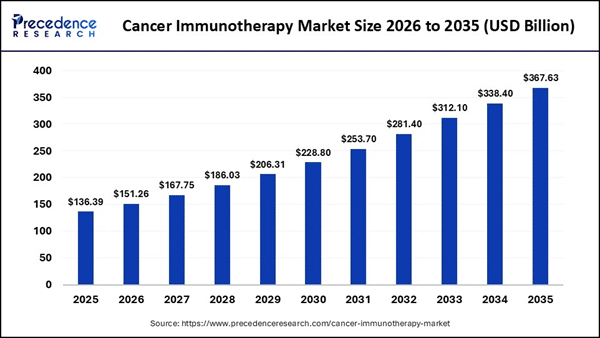
The broader cancer immunotherapy market was valued at approximately $136B in 2025 and is projected to reach roughly $367B by 2035, growing at a compound annual growth rate (CAGR) of approximately 10.4%. Within that broader landscape, the NK cell therapeutics segment is expanding even faster — valued at $3.2B in 2024 and forecast to reach $8.6B by 2033. (GTBP)’s GTB-5550 program targets a portion of the estimated $362B global solid tumor market, according to the company's January 2026 IND announcement. The upcoming Phase 1 basket trial covers up to seven potential metastatic cancer cohorts: castration-resistant prostate cancer, ovarian cancer, breast cancer, head and neck cancer, non-small cell lung cancer, pancreatic cancer, and bladder cancer. The TriKE® mechanism is also designed to avoid the most severe side effects associated with competing modalities. CAR-T therapies are widely associated with cytokine release syndrome (CRS) and neurological complications stemming from T-cell hyperactivation. GTBP's approach selectively activates NK cells rather than T cells, potentially offering a more tolerable safety profile — an important distinction in the immunotherapy landscape. Big pharmaceutical companies have taken notice: Sanofi and Gilead both completed significant NK cell engager deals valued at over $1B in milestone and upfront payments, according to GT Biopharma's March 2026 corporate presentation. Early Data Behind The Platform
(GTBP)’s clinical foundation is not speculative — the company's first-generation TriKE®, GTB-3550, completed a Phase 1 study in AML and MDS patients and demonstrated reproducible NK cell proliferation, activation, and persistence across all patients and all dose levels, with minimal clinically significant toxicity. In select patients, GTB-3550 showed reductions in CD33+ bone marrow blast levels: 33.3% in one patient, 61.7% in another, 63.6% in a third, and 50% in a fourth — encouraging early signals for a first-in-class modality at lower dose levels. Building on those results, (GTBP)’s second-generation candidate GTB-3650 incorporates camelid nanobody technology for improved potency, enhanced binding affinity, commercial manufacturing capabilities through Cytovance, and full proprietary ownership by GT Biopharma. The Phase 1 dose escalation trial (NCT06594445) is actively enrolling, with Cohort 5 dosing (25 µg/kg/day) expected to begin in Q2 2026 and the next data update anticipated in Q3 2026 — covering longer-term follow-up for patients in Cohorts 1 through 3, plus initial observations from Cohorts 4 and 5. (GTBP)’s Runway Into Q4 2026
According to (GTBP)’s full year 2025 financial results, the company held cash and cash equivalents of approximately $7M as of December 31, 2025, and an unaudited proforma cash balance of approximately $9M as of January 31, 2026. Management anticipates this runway to fund operations through Q4 2026 — spanning both the continued enrollment of the GTB-3650 Phase 1 trial and the planned mid-2026 initiation of the GTB-5550 basket trial. Full year 2025 R&D expenses came in at approximately $3.5M, down from $5.8M in 2024, primarily reflecting reduced production and material costs as the company sharpened its development focus. Why We’ll Have All Eyes On (GTBP) Tomorrow Morning
—Thursday, May 14, 2026 …1. FDA-Cleared Path: With GTB-5550 cleared by FDA for a planned mid-2026 Phase 1 basket study, (GTBP) has another clinical program approaching. 2. Recent Momentum: After an approximate 59% move in under a week, (GTBP) is now at the top of our watchlist. 3. Flying Under the Radar: With a market cap below $12M, (GTBP) sits in a smaller valuation range than many clinical-stage biotech names, which could suggest a higher-potential for growth. 4. Active Trial: The Phase 1 dose escalation study for GTB-3650 is actively enrolling, giving (GTBP) a live clinical timeline. 5. Platform Design: The TriKE® platform allows (GTBP) to adapt its NK cell engager approach across different tumor targets. 6. Funded Runway: Management expects current funding to support (GTBP) operations through Q4 2026. 7. Q3 Update: A GTB-3650 data update is anticipated in Q3 2026, giving (GTBP) a defined upcoming milestone. Don’t Leave (GTBP) Off Tomorrow’s Watchlist…

Between the recent momentum, active clinical enrollment, upcoming Q3 2026 data timeline, and a second FDA-cleared program preparing to enter the clinic, (GTBP) is beginning to draw attention at a time when several important developments appear to be lining up simultaneously. The company’s TriKE® platform also gives (GTBP) exposure across multiple disease areas, while current funding guidance through Q4 2026 provides visibility into the next stage of clinical execution. For a biotech still operating with a market cap below $12M, the setup is difficult to ignore. Heading into Thursday morning, (GTBP) is the only one we’ll be tracking. Spend a few minutes on (GTBP) tonight, so you’re ready for tomorrow. Our morning update may be in your inbox early—keep an eye out for it. Sincerely, Gary Silver
Managing Editor,
MarketCrux
|





