Thanks for signing up for DividendStocks.com! It's the daily newsletter built for dividend and income investors.
Before we can begin sending your daily updates, there’s one quick step left. Please confirm your subscription using the link below so our emails reach your inbox.
- Exelixis delivered a major EPS beat driven by strong Cabometyx demand, highlighting the company’s profitability and continued leadership in kidney cancer treatments.
- The biotech is transitioning to a multi-franchise oncology model, with zanzalintinib targeting colorectal cancer and representing a potential $5 billion peak-sales opportunity pending FDA review.
- Heavy R&D investment alongside share buybacks signals confidence in the pipeline, positioning Exelixis for sustained growth beyond its current single-drug revenue base.
- Special Report: [Sponsorship-Ad-6-Format3]
Exelixis Inc. (NASDAQ: EXEL) stock is down about 2% in early trading the day after the company delivered a solid, yet mixed earnings report. The company reported earnings per share (EPS) of 94 cents ($0.94), which was 27% above the consensus estimate and 95% higher year over year (YoY).
That profit showed up in the company's operating margin, which Exelixis plans to reinvest into research and development for its franchise strategy. The company also repurchased $264.5 million of its stock.
The revenue picture was mixed. The $598.66 million in revenue fell short of expectations of $609.17 million, although it was 5% above the $566.76 million reported in the same quarter last year. Revenue was largely driven by Cabometyx, the company's branded formulation of cabozantinib used across multiple cancer types.
Exelixis forecasts revenue between $2.52 billion and $2.62 billion for 2026. An important caveat: that outlook does not include potential revenue from zanzalintinib, the pipeline candidate for colorectal cancer, if it receives regulatory approval.
What Makes Exelixis Different?
On one level, Exelixis offers investors the same risk-reward profile as other biotech companies. But investors should take a closer look at the company's franchise strategy.
Exelixis is building comprehensive treatment ecosystems around specific drug molecules. The goal is to develop deep expertise in particular tumor types with multiple treatment lines and combination options that physicians can deploy at different stages.
Put simply, Exelixis is working to have multiple arrows in its quiver for specific cancers — first-line, second-line, and combination therapies — aiming to be the go-to choice for oncologists treating kidney, colorectal, and neuroendocrine cancers.
Two key takeaways from the fourth-quarter report:
- Cabozantinib is effective in kidney cancer both as monotherapy and in combination with immunotherapy. It is the primary driver of revenue today.
- Zanzalintinib is described as "the foundation of future oncology franchises," with the potential for up to $5 billion in peak annual sales.
Consolidation Now, Growth Later
At 18x trailing twelve-month (TTM) earnings and 21x forward earnings, EXEL stock commands a slight premium to the broader biotechnology sector. Exelixis's franchise model and deep pipeline support that premium as a bet on future growth.
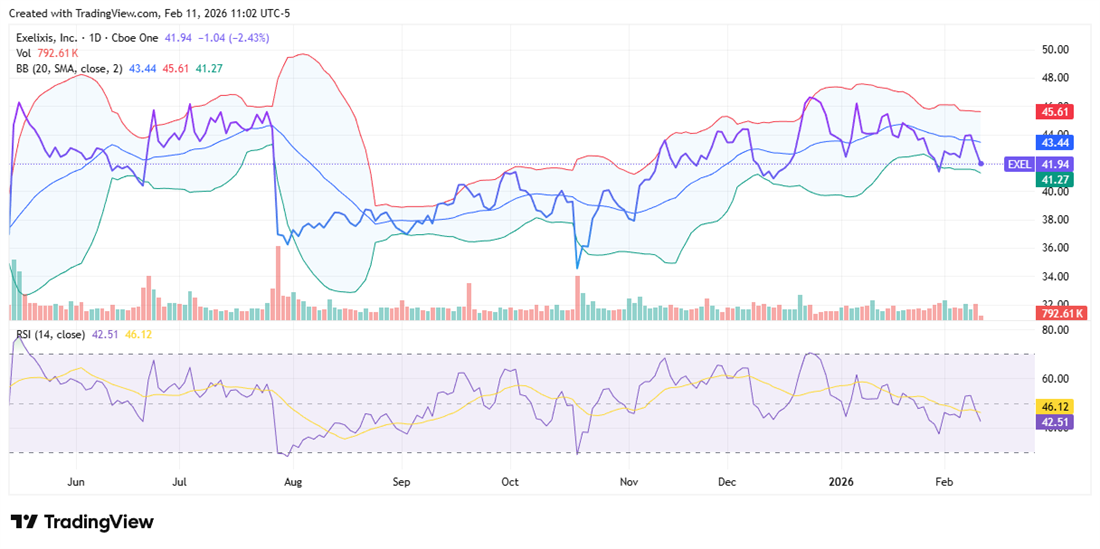
The EXEL chart looks constructive, with the stock sitting just below the 50-day simple moving average (SMA), which has recently acted as a key technical level. Momentum indicators were neutral heading into earnings; the stock was 8.6% below its consensus price target of $46.12.
After earnings, Wells Fargo reiterated an Equal Weight rating on EXEL and raised its price target to $35 from $30. Barclays also raised its price target to $44 from $41 on Feb. 4.
While EXEL is in a consolidation pattern for now, if the company's growth materializes, all-time highs could be within reach over the next 12 months.
 Exelixis Is at an Inflection Point
Exelixis Is at an Inflection Point
The story isn't just about beating earnings expectations or hitting revenue milestones. Exelixis is shifting from a single-product company to a multi-franchise oncology player, and 2026 looks like the year that transition becomes tangible.
The FDA decision on zanzalintinib in colorectal cancer (PDUFA date: Dec. 3, 2026) represents the company's first major expansion beyond cabozantinib. If approved, it would open the door to a potential $5 billion peak-sales opportunity and validate the franchise strategy the company is building.
The real indicator is R&D spending. Despite strong profitability, Exelixis is maintaining roughly $1 billion in annual R&D investment while also executing share buybacks — a sign of confidence in its pipeline. The company is balancing shareholder returns with aggressive development across seven pivotal trials for zanzalintinib alone, plus four early-stage programs advancing toward full development.
For context, the expanded GI sales team isn't just about net growth; it's pre-positioning for a potential zanzalintinib launch later this year. The pieces are being put in place for a different kind of biotech story: sustainable, multi-product growth anchored in deep tumor expertise rather than binary, single-drug bets.